Cavallette a pranzo? Tutta la verità sui #NovelFood https://t.co/iCWllghVz8 pic.twitter.com/rGAax5Vuj7
— Beppe Grillo (@beppe_grillo) October 31, 2015
Scientific and technological advances – especially in nanotechnology – are turning supermarkets into large modern bazars where any imaginable ingredient can be easily found. Due to globalization and free trade agreements, agricultural products from non-European countries are nowadays part of our daily allowance.

As an example of this food revolution is chia seeds: rich in omega-3 fatty acids, antioxidants, calcium and fiber. It is an ingredient that can be easily added to any kind of recipes (here is a list of some delicious examples). Originally from Mexico, chia seeds have successfully obtained the EU organic farming certificate as ‘novel food’.

Another, more controversial, example is spirulina, a dietary supplement available in tablet, flake or powder, which contains all essential proteins, carbohydrates and vitamins. It grows free of pesticides in China and has been rigorously tested before entering the EU market showing no signs of toxicity. However, experts cannot fully assert its health safety. Or stevia, the most widespread ‘novel food’, is a natural sweetener 150 times sweeter than sugar. It was banned in the United States last decade and brought back into legality years later. The EU approved its commercialization in 2011.
Although concerns on this matter started in 2008, when the term ‘novel food’ was not yet precisely defined, discrepancies between the European Council and the Parliament have postponed the reform process. One of the crucial issues was whether or not to include cloned animals’ products within the Novel Food Regulation. Even though the Commission decided to treat it as a separate matter in 2011, the non-official agreement approved last week highlights again the need of incorporating the cloning issue.
Thus, the creation of new organisms and the arrival of ‘unknown’ ones raise uncertainty in terms of public health. The fast-growing number of the so-called ‘novel food’ has provoked a public debate in the food industry calling for an urgent review of the current European Food Regulation, which is considered “complex, expensive and time-consuming”. For this reason, safety and proper labelling must be guaranteed before any of this new food get into the EU market.
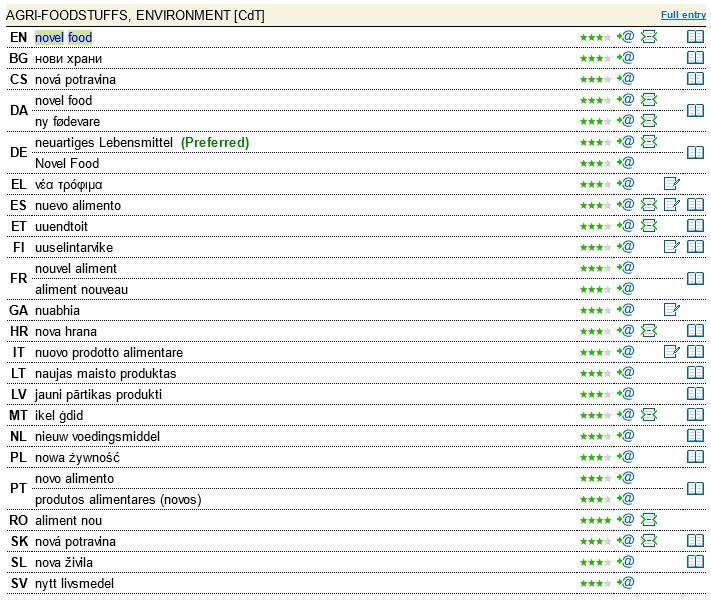
Today the definition of ‘novel food’ given by the European Commission is as follows: “food that has not been consumed to a significant degree by humans in the EU prior to 1997, when the first Regulation on novel food came into force. Novel food can be newly developed, innovative food or food produced using new technologies and production processes as well as food traditionally eaten outside of the EU.” (Also in IATE in all EU languages).
From now on, the approval of the new Novel Food Regulation by the EU will play an important role in major challenges of our time: growing world food consumption, environmental preservation and sustainable food production. What can broaden the horizons is to answer the question: what will we be eating in 2025?
Contribute to IATE! A terminologist for the respective language will revise your answer and decide whether to validate them. Given the implications of the process, a delay is to be expected.

Sources:
- European Commission . 2011. Novel food. [ONLINE] Available at: http://ec.europa.eu/food/safety/novel_food/index_en.htm. [Accessed 06 November 15].
- European Parliament. 2015. Updating rules on novel foods to keep up with scientific advances. [ONLINE] Available at: http://www.europarl.europa.eu/RegData/etudes/BRIE/2015/564383/EPRS_BRI%282015%29564383_EN.pdf. [Accessed 06 November 15].
- European Parliament. 2015. Novel Foods. [ONLINE] Available at: https://polcms.secure.europarl.europa.eu/cmsdata/upload/9c051023-d1a2-49e0-bd5f-87862c8974bf/Novel%20Foods.pdf. [Accessed 06 November 15].
- European Parliament. 2015. Novel Foods: food safety MEPs back deal with Council. [ONLINE] Available at: http://www.europarl.europa.eu/news/en/news-room/content/20150623IPR69710/html/Novel-Foods-food-safety-MEPs-back-deal-with-Council. [Accessed 06 November 15].
Written by Ana Escaso Moreno
Communication Trainee at TermCoord
Journalist & Social Media manager
